What is Point of Care Testing (POCT)?
Point of care testing is testing that is typically performed near the site of patient treatment. Point-of-care tests are less complex versions of tests performed by a standard hospital or reference laboratory. Due to its convenience, many different types of facilities utilize this form of testing, from schools to urgent care centers to hospital emergency rooms.
Point-of-care tests are easy to perform and do not require much technical training. Therefore, the range of people able to perform the testing is greatly expanded from college-educated and trained medical laboratory scientists. Basically, any person who has gone through the proper training and competency procedures for a particular point-of-care test can perform that test. It is not uncommon for medical assistants, CNAs, or x-ray techs to perform simple POCT tests in most medical facilities.
The purpose of POCT is for the clinician to make a change in a patient’s treatment in real-time. For example, a patient who is on blood thinners goes to the doctor because they notice increased bruising. In the office, the MA/nurse/doctor can perform a simple point-of-care test to determine the patient’s PT/INR. If these numbers are elevated, the physician will be able to quickly make alterations to the patient’s treatment and lower the dosage of blood thinner the patient is taking.
In fact, it doesn’t even need to be a medical professional that utilizes POCT. Diabetics do this several times a day when they check their blood sugar. If their sugar is too low, they may drink a glass of orange juice, and if it’s too high they know to take an extra insulin shot.
Difference Between Non-waived and Waived Testing
Testing that is performed at the point of care is considered waived testing. This section will explain the difference between waived (POCT) and moderately complex (standard lab) testing.
Waived Testing
Waived tests are tests that are considered to be low risk for producing incorrect results and that pose no reasonable harm to the patient if the test is performed incorrectly. These tests are cleared for home use. Tests are considered waived if they meet the requirements under FDA regulation 42 CFR 493.15(c). The most common example of one of these tests is a home glucometer used by a diabetic person. Laboratories that perform this kind of testing do not need to hold a CLIA certificate. These kinds of laboratories are most often seen in urgent care facilities, schools, or any other facility where non-laboratory personnel performing the tests.
Non-waived Testing
Non-waived testing refers to moderately or highly complex tests. These types of tests are performed in a standard hospital or reference laboratory. Labs performing these test must hold a CLIA certificate and trained medical laboratory scientists and technicians must perform these tests.
To determine if a test is a non-waived test, the FDA reviews the test’s product insert and rates the test using a criteria scorecard. There are seven (7) sets of criteria that the FDA uses to rate a test: knowledge; training and experience; reagents and materials preparation; characteristics of operational steps; calibration, quality control, and proficiency testing materials; test system troubleshooting and equipment maintenance, and; interpretation and judgement. Each set of criteria is given a rating of 1-3, with 1 being the least complex and 3 being the highest level of complexity. The scores for each of the seven criteria are then added together and, if the score is 12 or less, the test is considered moderately complex. If the score is above 12 then the test is considered highly complex.
Educational and Training Requirements
Typically, the most frequent users of POCT are nurses, however, any medical personnel who have been through the proper training can perform waived testing. Training and competency programs are designed by the laboratory Point-of-Care Coordinator (POCC) and approved by the laboratory director. The POCC must be a formally trained MLS/MT. The actual training of the staff using the POCT testing devices is done by nursing education in cooperation with the POCC.
For POC being implemented in a doctor’s office, a laboratory does not need to be involved. The doctor’s office just needs to be CLIA certified. A private practice physician office has to go through all the same validation, training, and competency requirements as a laboratory.
POCT education involves three steps consisting of initial training, certification, and competency.
Initial training: This is the step that introduces the staff to the new POC test/device. This is when the user learns basic steps of operation, troubleshooting techniques, QC, components of the device, how to report results, and what to do if there is a critical value. The nurse educator must demonstrate how to use the device and give the staff hands on experience.
Competency: This is the step where the staff proves they can confidently perform the test and understand the testing concept. Competency consists of users completing a written quiz to ensure understanding of testing concepts and procedures. Observation of users performing QC and running a sample patient is the final portion of competency. Once these steps are completed successfully, the user can become certified. After the initial training and competency, users are retested for competency after six months. After that, they are tested annually.
Certification: Certification occurs after initial training and competency are completed. The user is given a user ID and passcode for the device and is entered into the health system’s POCT management system as an official user.
Components of a POCT program
This section will briefly explain the important components of a POCT testing program. It is important that the POCT testing program remains organized and open lines of communication between each department involved are well established.
Organizational Requirements and Overview of Roles
Implementing point of care testing involves the cooperation of many different departments including the clinical laboratory, IT, biomedical engineering, materials management, and nursing.
Clinical Laboratory: The clinical laboratory is responsible for validating all new POCT devices, performing semi-annual correlations, and device troubleshooting. The laboratory POCC is responsible for keeping track of all competency requirements, certifications, and credentials, and works closely with nursing education to ensure these requirements are met. The POCC also works in a QA capacity, ensuring that all critical values are treated appropriately and all follow-up tests are completed.
IT: IT is responsible for ensuring that all POCT devices interface properly with the POC device management system. Devices can interface in two ways, depending on the technology of the device. The results can either upload into the device management system from anywhere there is a WiFi signal in the hospital or periodically through the device dock whenever the device is left to charge. Once the results are uploaded into the device management system, they then need to download into the patient’s medical record. IT is responsible for ensuring these two systems interface properly with each other.
Biomedical Engineering: Depending on the device and hospital policy, biomedical engineering may be involved in annual/semi-annual device maintenance. They are also responsible for making any installs or modifications that may be required for the device to function properly in a department.
Materials Management: Materials management is responsible for ensuring all supplies are ordered. Depending on the facility and volume of usage, materials management will ensure that standing orders are on time, supplies are distributed to proper departments, and new lots are not released until the lab has completed lot-to-lot verification.
Nursing: In POCT, nurses (or any other trained medical personnel) are the end-users of the point-of-care devices. Nurses are the staff members that are actually performing patient testing. They are the ones responsible for performing QC, daily maintenance, and basic troubleshooting procedures on the instrument when in use.
Policies and Procedures
Policies and Procedures are written by the laboratory in cooperation with the nursing department. Procedures are technical instructions that tell the users how to operate the device or perform the test. Procedures are usually written strictly by the laboratory and signed off by the medical director. Policies are broader in nature and deal with the overall process, including proper documentation of critical values, which follow-up tests should be ordered, how to document QC, etc. Policies require cooperation between both nursing and the laboratory staff.
Results reporting
Depending on the test being performed, results can be reported and recorded in a few different ways. In the case of an automated POC device, such as a glucometer, results are stored in the instrument and uploaded into the device management system, which then automatically cross over into the patient’s medical record. In the case of manual POC tests, such as a pregnancy test, results may be either directly entered into the patient’s MR or recorded on a log sheet to be entered at a later time. In the case of either testing situation, critical results and proper follow-up must be properly documented.
Quality Assurance
Quality assurance is a joint effort between the nursing department and the laboratory. While POCT offers quick results, results may not always be as accurate as a laboratory analyzer. Most critical values in POCT must be followed up with a second sample sent to the main hospital laboratory or a reference laboratory to confirm the critical value. The laboratory POCC is able to follow up on critical values via the device management system and laboratory information system (LIS) to ensure that samples are sent to the laboratory to confirm critical values. Any critical values that are not followed up properly will be addressed with the appropriate nurse manager. The laboratory POCC also ensures that all QC and maintenance is completed and documented properly.
Device Selection
POCT devices are chosen by the needs of the department where it will be used and the capabilities of the laboratory. The device has to make sense for the end user’s workflow and successfully integrate into information systems already in place in the health system and laboratory.
For example:
Let’s say a nursing home wants to implement a rapid antigen test. Nurse Jones has a friend that works in a nursing home across town that has already implemented a rapid antigen test. Nurse Jones, without consulting any other department, decides to buy the same rapid antigen testing kit as her friend’s facility. The particular test that Nurse Jones selected is cumbersome, has a lot of steps, and only one sample at a time can be processed. This particular test works fine for Nurse Jones’ friend because the nursing home she works at is small and has a low volume of samples. Nurse Jones, however, works at a large facility where implementing this sort of testing may be burdensome. When the POCC for the facility sees the test that Nurse Jones purchased, she informs her that there is a much easier and faster test that has comparable performance standards that could have been purchased instead.
In order to successfully select a device, it’s best to form a committee consisting of all key stakeholders mentioned in the above section. Each department has its own unique requirements and expertise that will lend itself to picking an appropriate device. In the above example, had Nurse Jones consulted with the POCC, she would have had a test more suited to her department’s needs.
Examples of POCT Instrumentation
The purpose of POCT is to produce immediate results that can affect patient care, treatment, and disease management. This section will look at three of the most common POC devices in use. Both of these devices can be used either in a clinical setting or in a patient’s home.
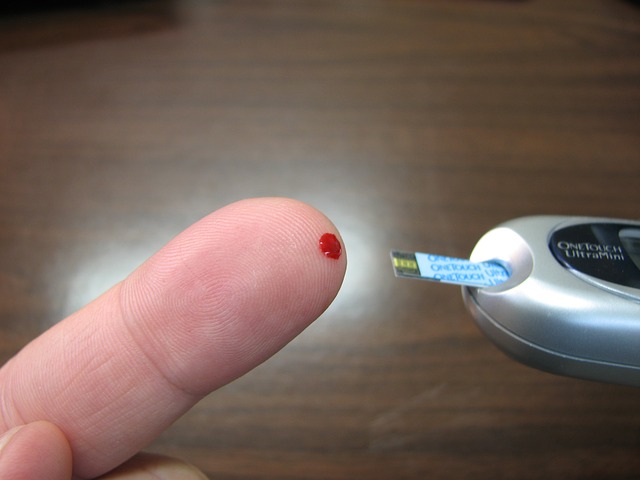
Glucometer
Glucometers are the POC device that immediately comes to mind when thinking about POCT. The purpose of these devices is to monitor a diabetic patient’s blood sugar. This device is easy to use and requires a small finger stick to obtain a drop of blood, which is transferred to a strip that is inserted into the device. Once inserted, the device will give a blood sugar reading in under a minute. The clinician (or patient) will be able to adjust their insulin and dietary routine accordingly. This device can be used either in a clinical setting or in a patient’s home.
Coagulation Device
These devices, while not as common as the glucometer, work much in the same way. They can be used by patients on anticoagulant therapy to monitor their dosage at home. Like the glucometer, a small amount of blood from a finger stick is applied to a strip in the device. In a few minutes, the instrument will give an INR reading allowing the patient to adjust their dosage for the day. This device can be used either in a clinical setting or in a patient’s home.
Portable Blood Gas Device
These devices are used solely in hospitals and typically found in the ED or ICU. These can be used on-site by a clinician for a patient that is in severe distress. While these devices are great for giving fast, on-the-spot results, the amount of sample used and the way the sample is collected may not give optimal results. Results should be confirmed by following up with a true blood gas that is sent to the laboratory.
Pros and Cons of POCT
Any laboratory test or process has pros and cons. POCT is no different. Let’s take a brief look at the pros and cons of POCT.
Pros
Fast TAT and small sample volume: The test is performed near the patient and much less invasive. Rather than performing a venipuncture, a small finger stick is all that is required. Test results are also available in minutes rather than hours.
Patient is not lost to follow up: The test is performed on-site and in front of the patient. If the test results are concerning the clinician can discuss treatment options with the patient right away. With traditional laboratory testing, results may not be available for 24 hours or more. By that point, the clinician may not be able to contact the patient to discuss follow-up treatments in a timely fashion.
Complex machinery not required: POCT can be performed on handheld or tabletop devices rather than large laboratory analyzers. The POC devices are easy to use and require few steps to complete the test
Cons
Expense: POC tests are much more expensive than traditional laboratory testing. For example, running a POC glucose test may cost $3.00 per test, whereas a serum glucose sample sent to the lab may cost 10 cents. While $3.00 may not seem like a lot, it adds. Consider a large health system that runs 100,000 POCT glucose tests per year.
Lack of control over process: Because POCT isn’t performed in the lab, there is little control over how testing is performed. The POCC relies on hundreds of users to follow protocol and procedures when performing testing in their own departments.
Lack of training: While POCT users and operators are required to be trained, certified, and competency tested, it’s hard for a POCC to keep track of this. POC devices have an automatic lockout when a user isn’t certified or has let their competency lapse, but manual tests are much harder to control.
Validation and Correlation
Even though most POCT are generally easier to perform than non-waived laboratory tests, they are still subject to the same validation requirements. Please see our blog entitled Laboratory Test Method Validation and Verification by Tracy Frankowski for an in-depth explanation of test method validation. Adequate validation ensures that the test produces the correct results within the performing facilities workflow.
POC devices also need to be correlated with each other and with standard laboratory machinery. For example, let’s say a facility uses two POC devices for coagulation studies. These two instruments need to be correlated to each other and with the coag instruments in the clinical laboratory. A typical correlation study is done using a sample with a known value. Depending on the test, contrived samples with known values can be used as well as patient samples with known values. This is to ensure that each instrument is giving consistent results. According to CAP regulations, this needs to be completed every six months.
Lack of standardization: Each department may have slightly different variations on how they perform procedures. In department A, the nurse manager may be very diligent, ensuring that all reagents are kept at proper temperature and devices are handled properly. In department B, the nurse manager may be more lax, allowing reagents to sit at room temperature for too long or not ensuring devices are handled properly. These things can affect results from department to department.