In Hemostasis – Part 2, we discussed fibrinolytic therapy and the use of fibrinolytic agents to treat emergency room patients and/or inpatients with acute, often life-threatening conditions such as STEMI, PE, DVT, stroke (CVA/TIA). In other words, these agents are used as “clot busters” for emergent cases of an active thrombus or embolus.
This final blog in the 3-part hemostasis series will discuss treatments that are more therapeutic in nature, used to prevent future thrombi or emboli in patients that have a history of blood clots. These treatments are also used before and/or after certain surgical procedures (e.g., artificial joint replacement).
We will also discuss the principles of diagnostic and therapeutic coagulation testing, as well as various disorders of hemostasis.
Therapeutic Anticoagulation Treatment (Anticoagulation Therapy)
Therapeutic anticoagulation treatments, commonly known by many people as “blood thinners”, are used most frequently for patients with a known history of blood clots or patients before/after certain surgical procedures. Examples of these therapeutic medications are listed below along with their mechanisms of action.
- Warfarin/Coumadin – oral Vitamin K inhibitors; patients must be frequently monitored with PT/INR blood testing so the medication dosage can be altered as necessary (to prevent excess bleeding and possible hemorrhage).
- Other oral drugs that do not typically require frequent blood testing (now more commonly used than warfarin/coumadin).
– Rivaroxaban (Xarelto) – inhibits free and stable clot-bound factor Xa.
– Apixaban (Eliquis) – inhibits free and stable clot-bound factor Xa and prothrombinase activity.
– Dabigatran (Pradaxa) – direct thrombin inhibitor.
– Edoxaban (Savaysa) – inhibits free factor Xa and prothrombinase activity; inhibits thrombin-induced platelet aggregation - LMWH (low-molecular-weight heparin) – binds to antithrombin and prevents activation of factor X; more commonly used; subcutaneous injection – dalteparin, enoxaparin (Lovenox), tinzaparin – used to treat and prevent DVT and PE in patients with previous DVT/PE or patients more prone to these (pregnancy, following certain surgeries, patients undergoing chemotherapy treatments, patients with acute cardiac syndrome and heart attacks).
- Aspirin – inhibits platelet aggregation.
- Plavix (clopidogrel bisulfate) – inhibits platelet aggregation; often used to treat patients with myocardial infarction (MI), ischemic stroke, peripheral vascular disease, or acute coronary disease.
- Synthetic Heparin-like drugs (Fondaparinux – subcutaneous injection)
Diagnostic and Therapeutic Coagulation Testing
Some tests are diagnostic and others therapeutic. Diagnostic testing would likely be done before surgery to assess your blood’s ability to clot, to help diagnose liver problems, or to determine what is causing a patient to develop blood clots or to hemorrhage. Therapeutic testing would be done to monitor the effectiveness of certain anticoagulation medications (“blood thinners”), most commonly warfarin (coumadin).
The most commonly ordered coagulation tests are the platelet count, prothrombin time (PT), and activated partial thromboplastin time (aPTT or PTT). It is these tests that will be discussed in the most detail in this blog. Other coagulation tests will be mentioned with only brief descriptions and the overall purpose of each test.
Platelet count, PT/INR and PTT tests are used as screening tools to determine whether a patient has a coagulation problem and are often ordered before any surgical procedure. A physician may also order these tests if a patient has signs and symptoms of a bleeding disorder, such as frequent nose bleeds, unexplained bruising, etc. They are also helpful diagnostic tests ordered on patients that present with signs and symptoms of potential bleeding issues, such as hematemesis, hemoptysis, or vaginal hemorrhaging.
Platelet count (PLT) – The reference range for a platelet count is 150,000 – 400,000 per microliter (µl) of blood. High platelet counts do not typically cause issues. However, low platelet counts (below 150,000/µl) may cause abnormal bleeding from minor scrapes or cuts, excessive bruising, frequent nose bleeds, etc. Low platelet counts can be caused because the bone marrow is not producing enough platelets or because something in the body destroys platelets faster than the bone marrow can produce them.
Having a platelet count within the normal reference range does not necessarily mean a patient is free of platelet issues. Relatively early in my career as a medical technologist, I encountered a patient who had a platelet count >1,000,000/µl, yet had multiple unexplained bruises and uncontrolled bleeding even with minor scrapes or cuts. Her physician ordered a bleeding time (a test no longer performed), which was extremely extended (much greater than the upper reference range). Although she had such a high platelet count, her platelets were not functioning properly, as determined by further platelet function testing.
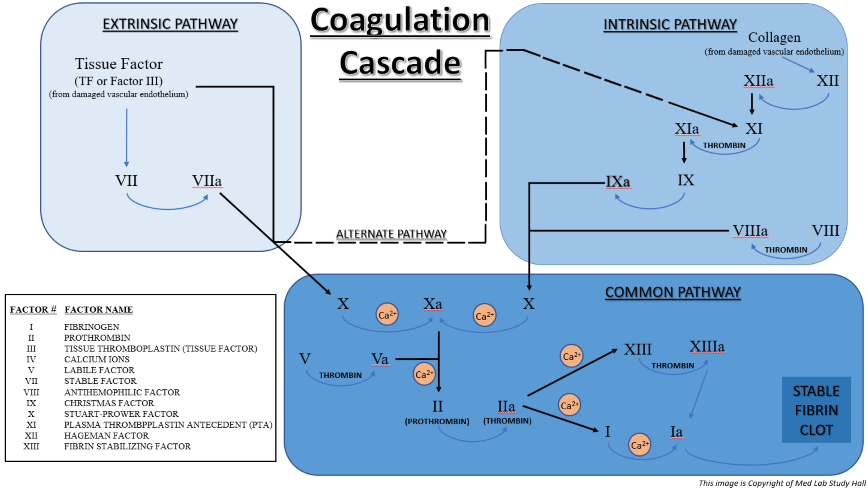
Prothrombin Time (PT) – This test determines the functionality of the patient’s extrinsic and common coagulation pathways, factors VII, X, V, II (prothrombin), I (fibrinogen). It is used to help diagnose clotting disorders, as well as to help monitor patients taking certain therapeutic coagulation medications.
A blood sample is collected in a sodium citrate collection tube. The sodium citrate in the tube binds up calcium (Ca++) in the blood sample, which prevents the sample from clotting. It is crucial to properly fill this tube, as the ratio of blood to sodium citrate anticoagulant must be 9:1 for the PT test to be accurate. The blood sample is centrifuged to separate the plasma from the blood cells. This plasma is considered citrated plasma.
The PT testing process is as follows.
- Patient citrated plasma and PT reagent (which contains both thromboplastin and CaCl2) are prewarmed separately at 37⁰C for 1-2 minutes to replicate normal body temperature and the process that would actually take place in vivo. The source of the thromboplastin (TF) is derived from homogenized brain tissue. (Ca++ is needed for clotting to occur, so CaCl2 is present in the PT reagent to replace the Ca++ that was bound by the sodium citrate in the blood collection tube.)
- Specific aliquots of prewarmed patient plasma and prewarmed PT reagent are combined in a testing cuvette.
- From the time PT reagent is added to the patient’s plasma, the test is timed until the plasma clots. Clotting indicates that prothrombin has been converted to thrombin. This time is reported out in seconds as the Prothrombin Time (PT).
Depending on the source of the thromboplastin used, a PT within 10-15 seconds is considered a normal diagnostic level. If levels are below this range, it means your blood is clotting more quickly than normal, which could be caused by:
- vitamin K containing supplements,
- medications containing estrogen, such as hormone replacement therapy or birth control pills, or
- increased consumption of foods that are high in vitamin K (soybean products, green tea, liver, chickpeas, broccoli, Brussel sprouts, cauliflower, cabbage, and leafy green vegetables such as kale, Swiss chard, spinach, collard greens, mustard greens, etc.).
If levels are above the normal diagnostic level, it means your blood is clotting more slowly than normal, which may cause potential bleeding issues. This may be caused by:
- liver problems,
- inadequate levels of factors VII, X, V, II (prothrombin), and/or I (fibrinogen),
- anticoagulation therapy (“blood thinners”)
Internationalized Normalized Ratio (INR) – This test is a calculated value that is determined from a patient’s PT value that adjusts for changes in PT reagents used and allows for comparison of results from different laboratories. It is automatically reported out along with the PT result.
The INR therapeutic range for patients on warfarin/coumadin is generally 2.0-3.0. The result of this test is only important for patients that are taking warfarin/coumadin for anticoagulation therapy. Physicians use the INR results to determine whether a patient needs to maintain, increase, or decrease their dosage of warfarin/coumadin.
Activated Partial Thromboplastin Time (aPTT or PTT) – This test determines the functionality of a patient’s intrinsic and common pathway clotting systems, evaluating factors I (fibrinogen), II (prothrombin), V, VIII, IX, X, XI, and XII. It is used to help diagnose clotting disorders, as well as to help monitor patients on heparin therapy.
The blood sample used is the same as that used for the PT (see above).
The PTT testing process is as follows.
- Specific aliquots of patient citrated plasma and PTT reagent are combined in a testing cuvette and incubated at 37⁰C for 1-2 minutes to replicate normal body temperature and the process that would actually take place in vivo. PTT reagent contains both an activator (typically kaolin, silica, or elagic acid) and phospholipid (partial thromboplastin).
- CaCl2 is prewarmed separately at 37⁰C. (Ca++ is needed for clotting to occur to replace the Ca++ that was bound by the sodium citrate in the blood collection tube.)
- Prewarmed CaCl2 is added to the combined patient plasma/PTT reagent.
- From the time CaCl2 is added to the combined patient plasma/PTT reagent, the test is timed until the plasma clots. This time is reported out in seconds as the activated Partial Thromboplastin Time (aPTT or PTT).
A normal PTT usually ranges between 25-35 seconds, but ranges can vary based on laboratory.
The PTT is the most commonly used test to monitor heparin therapy. The therapeutic range for a patient on heparin therapy is approximately 1.5 to 2.5 times the mean normal value (roughly 45-75 seconds). The heparin dosage is adjusted by the physician based on these results.
Things that Interfere with PT/INR and PTT Testing
- Patients taking antihistamines, ascorbic acid, and/or salicylates may have prolonged results.
- Incorrect blood-to-citrate ratio; if the ratio is <9:1 the results will be prolonged (less Ca++, so results would be increased); if the ratio is >9:1 the results will be decreased (more Ca++, so results would be decreased).
- If the patient’s hematocrit that is >55%, the ratio of plasma:anticoagulant will be decreased and thereby yield prolonged results.
- Blood samples drawn from heparin lock, heparinized catheter, or from a patient receiving a heparin IV will prolong PTT results. If possible, have an RN turn off the heparin IV for at least two (2) minutes before collecting the patient’s blood. Then, collect a waste tube (a tube with no additive or another sodium citrate tube) before collecting the PTT specimen. Discard the waste tube. Also, make sure you notify the RN that you are done collecting the patient’s blood ASAP so the patient IV can be turned back on.
Interpretation of PT and PTT Results – A Recap
For diagnostic purposes, the PT is commonly ordered in conjunction with a PTT in order to better clarify any coagulation defects.
| PT RESULT | PTT RESULT | INTERPRETATION/POTENTIAL CONDITIO.NS |
|---|---|---|
| ↑ | Normal | Decreased or defective extrinsic pathway factor (VII); chronic low-grade DIC; liver disease; vitamin K insufficiency; warfarin/coumadin therapy. |
| Normal | ↑ | Decreased or defective intrinsic pathway factors (VIII, IX, XI, XII); presence of lupus anticoagulant; autoantibody against a specific intrinsic pathway factor; vWD. |
| ↑ | ↑ | Decreased or defective common pathway factors (I, II, V, or X; acute DIC; severe liver disease; warfarin/coumadin overdose. |
| Normal | Normal to slightly ↑ | May indicate normal hemostasis, but could potentially have normal PT/PTT results with mild vWF, mild coagulation factor deficiencies, or presence of weak lupus anticoagulant. |
Other Coagulation Testing
Factor V Liden – blood test ordered for patients believed to have inappropriate blood clot (thrombus) formation, such as DVT or venus thromboembolism (VTE). Click here for more information.
Fibrinogen – assesses the activity of fibrinogen; measures the time it takes for fibrin clot formation after adding thrombin to patient plasma; the addition of thrombin bypasses all other coagulation factors and focuses strictly on the function of fibrinogen. Click here for more information.
Lupus Anticoagulant Testing – a series of tests that aid in the evaluation of prolonged PTT results. Click here for more details.
Mixing Studies – patient plasma is mixed with a normal control plasma to determine whether a patient’s PT or PTT is due to a factor inhibitor or a factor deficiency. If the mixed plasma result normalizes, the prolonged patient result is due to a factor deficiency. If it does not normalize, it is due to a factor inhibitor.
Disorders of Primary and Secondary Hemostasis
Primary Hemostasis Disorders (caused by platelet defects, vWF, or receptor interference)
- Bernard-Soulier syndrome – deficiency of a platelet receptor that causes a defect in platelet aggregation; impairs the ability of platelets to bind vWF. Characterized by thrombocytopenia and large platelets.
- Glanzmann thrombasthenia – deficiency of a platelet receptor that causes defective platelet aggregation; patients have normal platelet counts.
- von Willebrand disease (VWD) – defect or deficiency in von Willebrand factor (vWF). Causes defects in platelet aggregation and deficiency of Factor VIII (since vWF is also a carrier of Factor VIII). There are different types of VWD: Type 1 is a partial quantitative defect or vWF; Type 2 has a normal quantity of vWF but has functional defect; Type 3 has complete absence of vWF.
- Immune Thrombocytopenic Purpura (ITP) – caused by the attack and destruction of platelets by the immune system.
Secondary Hemostasis Disorders (caused by clotting factor defects)
- Anti-phospholipid antibody syndrome – a rare, autoimmune disorder where the body’s immune system forms antibodies that attack phospholipids; leads to an increased risk of thrombosis (DVT, strokes, recurrent miscarriages in pregnant women, etc.)
- Disseminated Intravascular Coagulation (DIC) – a rare, serious condition, caused by another disease or condition, that causes an overactive blood clotting process; once all innate blood clotting factors are used up in this overactive clotting process, severe bleeding occurs. This condition is often fatal.
- Factor V Leiden – inherited disorder that causes a genetic mutation of the Factor V gene, resulting in thrombophilia (increased propensity to develop DVTs).
- Hemophilia A – genetically inherited deficiency of Factor VIII; predominantly occurs in males; treatment is recombinant factor VIII.
- Hemophilia B – genetically inherited deficiency or Factor IX; predominantly occurs in males; treatment is recombinant factor IX.
- Hemophilia C – mild form of hemophilia caused by a deficiency of Factor XI; predominantly occurs in Ashkenazi Jews (both sexes).
- Liver disease – since the majority of coagulation factors are synthesized in the liver, liver disease affects the production of these factors, thereby causing potential bleeding disorders.
- Protein C and S deficiencies – these proteins are needed to regulate clot formation, so deficiency of either (or both) can cause excessive clotting disorders.
- Vit K deficiency – Vitamin K is needed for the synthesis of coagulation factors II, VII, IX, and X; deficiency can cause severe bleeding issues.
In Summary
We covered a lot of information in our three-part series on hemostasis. Some information was more detailed than others. Where we felt applicable, we tried to provide links to other websites with additional information. Hopefully, you found our information helpful. If you haven’t done so already, please make sure to view Part 1 and Part 2 of our hemostasis series.

Videos to this author. Amazing collection of information that is clearly presented and a great guide for anyone interested in laboratory medicine. Congratulations on a job well done !
Thank you so much, Loretta! Your professional viewpoint and support for this endeavor has meant the world to me. 😊
Nice job, Dawn! I will be sharing this series with my students.
Tracy – Thanks for your “thumbs up” comment on the Hemostasis series and for all of your help with Med Lab Study Hall!